PHOTO : Courtesy MSF
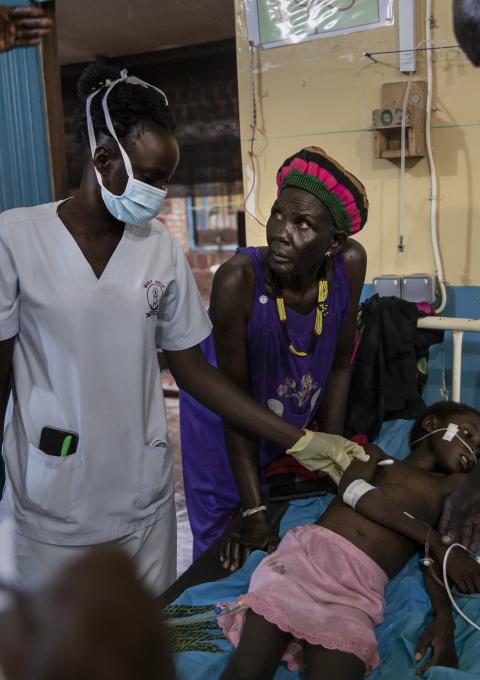
Médecins Sans Frontières (MSF) has completed the first full round of the R21 malaria vaccine in a refugee camp, marking a global first in the fight against one of Africa’s deadliest diseases.
The vaccination campaign took place at Kule refugee camp in Ethiopia’s Gambella region, home to more than 55,000 South Sudanese refugees, many of them children at high risk of severe malaria. MSF said the achievement shows that advanced vaccination programmes can be delivered successfully even in humanitarian and displacement settings.
“Completing a full malaria vaccination schedule in a refugee camp shows that lifesaving innovations can reach people who are often left behind,” MSF said, calling the rollout a major step forward for malaria prevention in emergencies.
The R21 vaccine was given to young children, the group most vulnerable to malaria-related illness and death. Health workers administered the final doses after months of follow-up, overcoming challenges linked to mobility, limited access, and constrained health infrastructure in the camp.
According to MSF, malaria remains one of the leading causes of illness in Gambella, where seasonal flooding and stagnant water create ideal breeding grounds for mosquitoes.
“Refugee children face overlapping risks: malnutrition, poor shelter, and limited access to healthcare which make malaria particularly dangerous,” MSF noted, adding that vaccines are critical alongside bed nets, treatment, and vector control.
MSF said the success of the Kule campaign could pave the way for wider use of the R21 vaccine in other high-risk humanitarian settings across Africa. Health experts have welcomed the development, emphasizing the importance of including displaced populations in global vaccination efforts as malaria continues to claim hundreds of thousands of lives each year.
Malaria, caused by Plasmodium parasites transmitted through mosquito bites, remains a leading cause of illness and death in many parts of Africa, particularly among children under five. Symptoms range from fever and fatigue to severe complications, including organ failure and death if untreated.
Vaccines aim to train the immune system to recognize and fight the parasite before it causes serious illness. Developing malaria vaccines is challenging because the parasite has multiple stages in its life cycle, making it harder to target than viruses or bacteria.
One of the first widely recognized malaria vaccines is RTS,S/AS01 (Mosquirix), developed by GlaxoSmithKline and approved by the World Health Organization in 2021. Given in four doses to young children, it reduces severe malaria cases by about one-third. While this marked a milestone, scientists continued seeking more effective solutions.
The R21/Matrix-M vaccine is the latest breakthrough. Developed by the University of Oxford in partnership with Novavax, early trials show R21 can provide up to 77% protection in children over 12 months, higher than RTS,S. Like other malaria vaccines, it requires multiple doses for full protection, with booster doses to extend immunity.
R21 is now being tested in real-world conditions, including refugee camps like Ethiopia’s Kule camp, demonstrating that even the most vulnerable populations can be reached. Experts stress that vaccines must complement existing malaria control tools such as insecticide-treated bed nets, indoor spraying, and preventive medicines to dramatically reduce infections and deaths and move closer to controlling this deadly disease.


